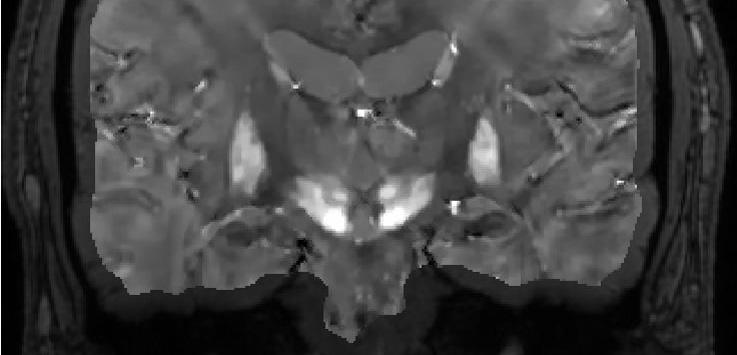
The long-term objective of this project is to develop noninvasive, robust, sensitive and accurate midbrain iron mapping for Parkinson's disease (PD). PD is a neurodegenerative disorder characterized by dopaminergic neuron loss in the substantia nigra pars compactor (SNc) and consequent motor disorders. While the neurodegenerative processes in PD may be multifactorial, prooxidant iron elevation in the SNc is evidently an invariable feature of both sporadic and familial PD forms, contributing to oxidative stress and mitochondrial dysfuction, and presenting as a tractable target for a disease modifying therapy. Therefore, noninvasive quantitative nigral iron mapping would be useful for diagnosing PD, assessing PD progression, and monitoring PD therapy. Noninvasive magnetic resonance imaging (MRI) is regarded as the most sensitive method for detecting small amounts of highly paramagnetic iron in midbrain tissue. The lab has developed quantitative susceptibility mapping (QSM), which enables a quantitative extraction of tissue magnetic susceptibility from gradient echo MRI data by deconvolving phase data with a dipole kernel. Estimation of iron from magnetic susceptibility must account for contributions of calcification, the other major susceptibility source in basal ganglia that can also be estimated using a recently developed ultrashort echo time MRI technique. Accordingly, the lab proposes to develop noninvasive accurate midbrain iron mapping using the QSM approach with the following specific aims. Aim 1: Develop noninvasive and accurate midbrain iron mapping based on QSM. Aim 2: Validate noninvasive measurement of substantia nigra iron using elemental analysis and immunohistochemistry. Aim 3: Establish that QSM is more sensitive than R2* for nigral iron mapping in monitoring PD iron chelation therapy.