Next-Generation Quantitative Acoustic Microscopy for Biomedical Applications
This technology-development project will significantly advance quantitative acoustic microscopy (QAM) systems and ultimately pave the way toward wide acceptance of the technology. QAM systems permit formation of maps of the acoustical and mechanical properties of tissues at microscopic resolutions.
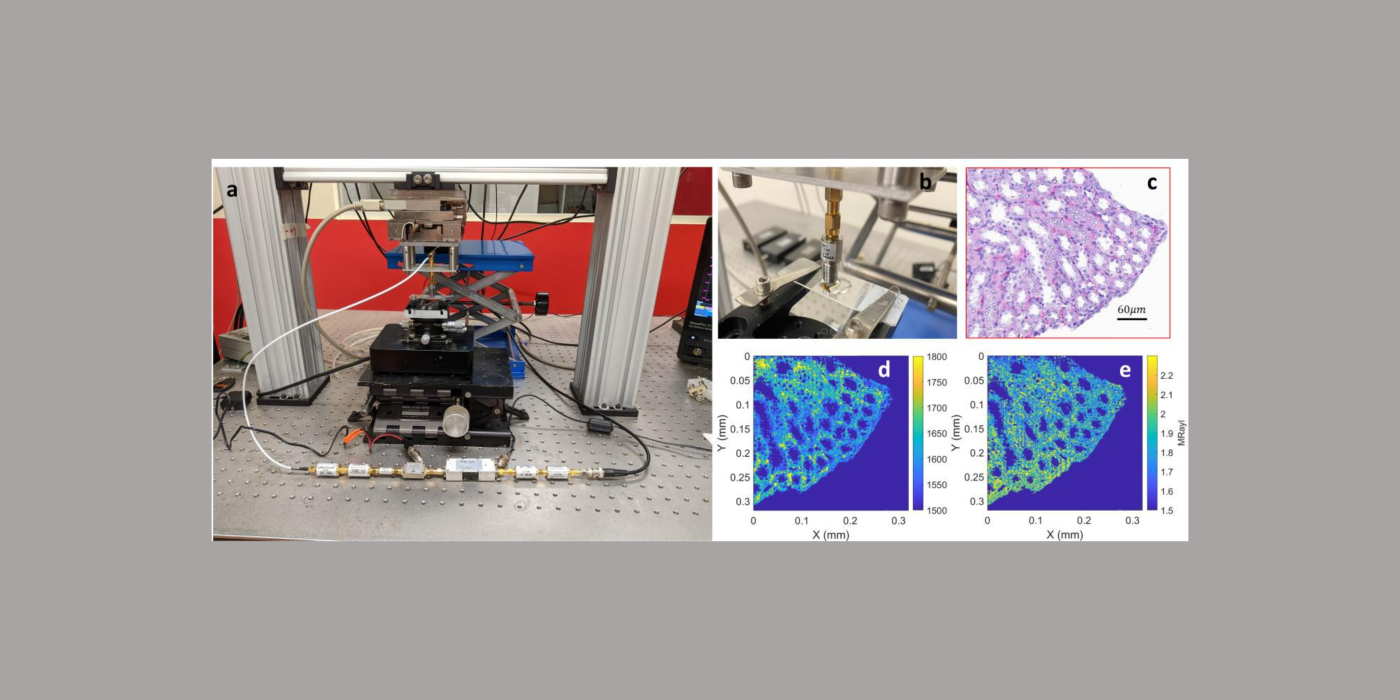
In collaboration with the University of Bristol (Bristol, United Kingdom), and the University of Lyon (Lyon, France), the Mamou lab will develop the next generation of QAM systems. Specifically, data science and coded-excitation approaches will be applied for the first time to QAM technology to yield better image quality, decreased scanning time, and greater ease of use. It will help usher in a new generation of novel, low-cost, user-friendly QAM instruments. QAM permits formation of fine-resolution (i.e., <7 µm at 250 MHz) maps of acoustic and mechanical properties of tissue sections that are <12 µm in thickness. These data can have great value in numerous preclinical investigations. Such property maps are not currently obtainable by any other microscopic-imaging modality, and the new generation of QAM technology—made possible by success in this proposed project—could become widespread in research laboratories and microscopy suites in commercial as well as academic research environments. Such new-generation QAM instruments could be used by technicians with limited knowledge of QAM and, in many ways, their use would be no more complicated than that of a conventional bright-field microscope. Importantly, these novel approaches to QAM will be demonstrated using already available resolution targets, phantoms, and biological tissues (ocular-tissue samples from a guinea-pig model of myopia and cancerous human lymph nodes). During the course of this project, optimal methods will be incorporated in a prototype QAM instrument capable of producing ultra-fine spatial resolution (< 2 µm) images much faster (<1 min) and for a much lower cost than current state-of-the-art QAM systems. In addition, use will be ``turn-key'' (i.e., requiring no technical knowledge and less than one hour of training.)
In-vivo ultrasound-based point-of-care instrument to assess myopia level and progression
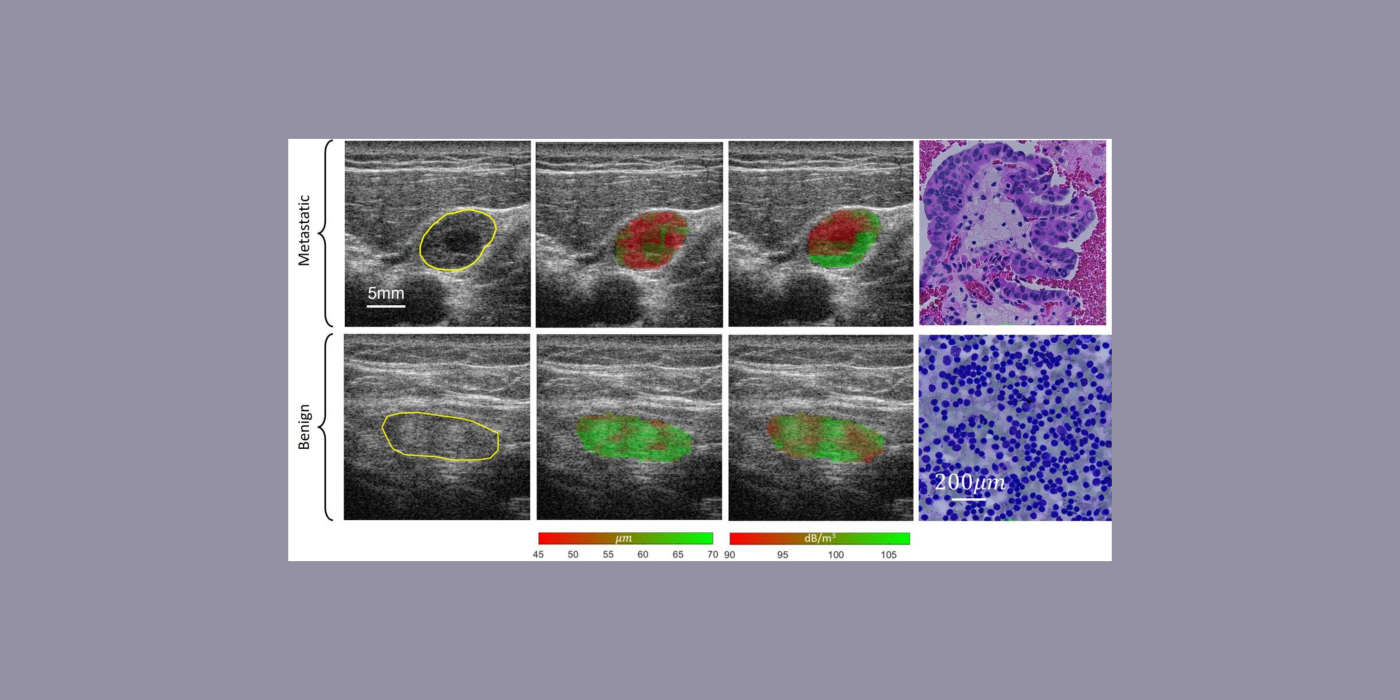
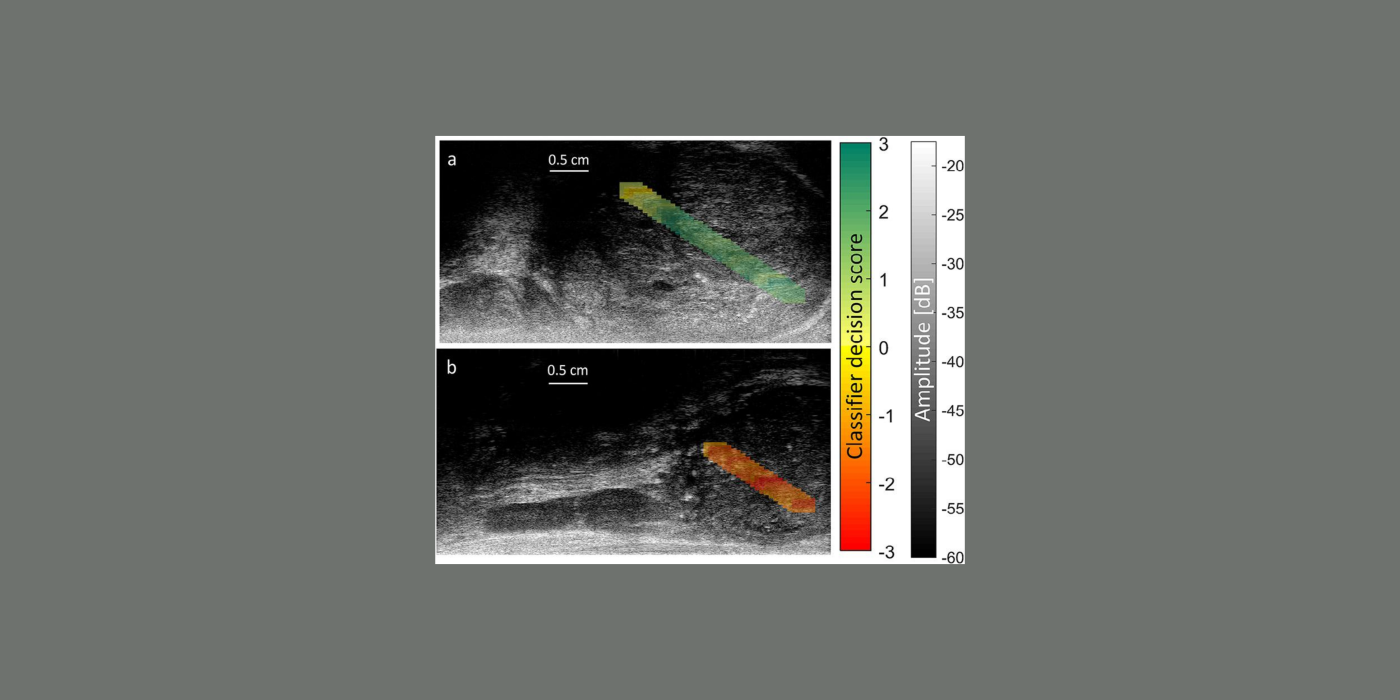
The goal of this project is to develop a novel, low-cost, compact, point-of-care (POC) ultrasound system to assess myopia severity and progression in vivo. Myopia prevalence is increasing worldwide and pathologic myopia is one of the leading causes of blindness. Myopia originates from structural and mechanical changes in the sclera that precede vision-threatening ocular changes. Currently, no method exists to assess myopia progression and severity quantitatively in vivo, and this project seeks to fill this gap. The POC instrument consists of a single-element, spherically focused transducer operating at an 80-MHz center frequency mounted in a handheld probe and connected to a computer equipped with dedicated software, hardware, and a pedal for ease of use implementing a graphical user interface. The POC instrument collects RF, A-line data at a high pulse-repetition frequency while in direct contact with the anterior sclera. The radiofrequency (RF) data is processed using quantitative (QUS) methods to derive so-called QUS parameters associated with the elastic and microstructural properties of the sclera. The QUS parameters are based on the backscatter coefficient and envelope statistics as well as on highly innovative passive elastography. The POC instrument is portable, low-cost, real-time, and nonionizing. It is similar to pachymeters currently used by ophthalmologists for biometric measurements, which will facilitate its adoption by clinicians. Currently, the POC instrument is deployed at our collaborating clinical site, the Singapore Eye Research Institute, to acquire and process data from 100 patients during a prospective clinical study aimed at assessing the value of the POC instrument for evaluating high-myopia patients. Specifically, the clinical study tests the ability of the system to predict myopia severity and progression. If successful, these studies will significantly advance myopia knowledge. Equally importantly, the proposed POC instrument will revolutionize care for myopia patients by providing a low-cost, easy-to-use, and safe means to assess disease severity and progression.
Adrian Basarab, Ph.D.
Alin Achim, Ph.D.
Marie Muller. Ph.D.
Stefan Catheline, Ph.D.
Kimberly Showalter Lakin, M.D.
Ronald Silverman, Ph.D.
Donny Hoang, M.D.
Carolyn Bayer, Ph.D.
Jerry Sebag, M.D.
Tadashi Yamaguchi, Ph.D.
Maoxin Wu, M.D., Ph.D.
Kirk Wallace, Ph.D.